Dr. SANTOSH MAHAPATRA
Dr.Gayatri Kanungo
Abstract
Multi-Characteristic Opsin (MCO) is an engineered opsin that can be used to photosensitize bipolar cells with the potential for greater spatial resolution at ambient light levels, thereby avoiding the need for an external amplifying device and associated phototoxicity.
AAV2 was used to deliver MCO-010 in advanced retinitis pigmentosa subjects. Safety and exploratory efficacy of intravitreal AAV2-MCO-010 dose escalation were evaluated to identify a safe dose for AAV2 based MCO-010 delivery. AAV2-vMCO-010 was well tolerated with no reported SAE at 16 weeks. Furthermore, exploratory endpoints demonstrated improvement in vision and visual function in retinitis pigmentosa subjects following a single dose.
AAV2-vMCO-010 is well tolerated with no serious adverse events. Higher dose AAV2-vMCO-010 appears to improve visual acuity and visual function compared to baseline measurements at 16 weeks and demonstrates tremendous promise in restoring vision for RP patients
Full Text
Abstract
Purpose: Optogenetics therapy offers the potential for vision restoration in patients with photoreceptor degeneration. Through the delivery of opsin encoding genes, residual retinal neurons take on the photosensitizing function of the photoreceptors. Such an approach focuses on disease phenotype versus a specific genotype deficit, therefore applicable to a wide patient population. Existing optogenetic tools utilize opsins that do not generate adequate electrical current in ambient light requiring an external device for stimulation. Hence, this study aims at targeting bipolar cells modifying them to be ambient light activable photoreceptor cell by intravitreal introduction of multi characteristic optin carried by Adeno Associated Virus 2 (AAV2) vector.
Methods: Multi-Characteristic Opsin (MCO) is an engineered opsin that can be used to photosensitize higher order retinal cells with the potential for greater spatial resolution at ambient light levels, thereby avoiding the need for an external amplifying device and associated phototoxicity. AAV2 was used to deliver MCO1 in advanced retinitis pigmentosa subjects. Subjects received prophylactic oral steroids prior to a single intravitreal injection of AAV2-MCO1 (vMCO-l). Safety and exploratory efficacy of intravitreal vMCO-l dose escalation were evaluated to identify a safe dose.
Results: vMCO-l was well tolerated with no reported serious adverse events at end of the study at 16 weeks. Ocular adverse events were limited to inflammation and rise in intraocular pressure that were controlled with topical medications. Furthermore, exploratory endpoints demonstrated improvement in vision and visual function in retinitis pigmentosa subjects following a single dose.
Conclusions: vMCO-l is well tolerated with no serious adverse events. Higher dose vMCO-l appears to improve visual acuity and visual function at 16 weeks compared to baseline measurements and demonstrates tremendous promise in restoring vision for retinitis pigmentosa patients.
Introduction :
In retinal degenerative diseases such as Retinitis Pigmentosa (RP), Stargardt’s disease, Leber’s congenital amaurosis (LCA) and Cone-rod Dystrophy, the photoreceptors that are responsible for conversion of light into electro-chemical signals, are degenerated. This prevents the generation of photo-induced signals in retina, breaking the vision-sensory related cascade of events within the visual system. Loss of photoreceptor cells and/or loss of photoreceptor cell function are the primary causes of reduced light sensitivity and blindness in advanced stage of retinal dystrophy.
Retinitis Pigmentosa (RP) refers to disorders characterized by degeneration of photoreceptors in the eye, which hinders visual ability by non-functional neuronal activation, and transmission of signals to the visual cortex (1-5). The prevalence of RP is approximately 100,000 patients in the US, out of which ~50,000 patients have advanced retinal dystrophy. Assuming the report of 1 in every 3000 people has RP, India has RP patient population > 460,000. RP is most often inherited as an autosomal recessive trait with large number of cases having this form of inheritance (3, 6, 7). Further, the degree of visual loss increases with ageing (8) and this is a major concern for our demographic changes towards elderly population. In some people, RP advances so slowly that vision loss does not occur for a long time. In others, the disease progresses faster and may lead to a loss of vision in one or both eyes. Most of the current clinical treatments are primarily focused on slowing down the progression of the disease (9), as there is neither a cure that can stop the degeneration (10) nor a therapy, other than retinal prostheses, that can restore vision lost due to the degeneration (11). Partial restoration of vision involves invasive surgical procedure for retinal implants (12). Two different types of retinal implants are being developed: subretinal and epiretinal implants (13). The subretinal implants are positioned in the area of the retina where the photoreceptor cells reside, between the pigmented epithelium and the bipolar cells (14). These retinal prostheses have been successful in generating visual perception in blind subjects (15-17). The disadvantages of using such subretinal implants include (i) chronic damage of the implanted electrodes, and (ii) insufficient current produced by microphotodiode from the ambient light to stimulate adjacent neurons (18, 19). The epiretinal implants are placed in the area of the retinal ganglion cells (RGCs) and the device functions by stimulating the RGCs in response to input obtained from a camera that is placed outside of the eye or within an intraocular lens (19, 20). The disadvantages of epiretinal implants include (i) cellular outgrowth due to surgical implantation, and (ii) disordered stimulation pattern resulting from the electrical stimulation of both the axons and cell bodies of the RGCs (19). Besides being invasive in nature, these methods for restoration of vision in blind patients are based on non-specific cellular activation and have low spatial resolution due to low number of electrodes (higher number or density of electrodes requires more power, leading to damage of neural tissue by heat), and hence able to improve vision with low spatial resolution.
In advanced stages of retinal degenerative diseases such as RP, the photoreceptors that are responsible for conversion of light into electro-chemical signals are degenerated. This prevents the generation of photo-induced signals in retina, breaking the vision-sensory related cascade of events within the visual system. There is no cure for these diseases, especially in the advanced stages. Since higher order neurons are still intact in degenerated retina, several stimulation methods target the higher order neurons, e.g. Bipolar cells and retinal Ganglion cells, which carry the visual information to the visual cortex. While direct electrical stimulation approaches require mechanical contact of electrodes to the retinal cells, indirect stimulation approaches such as optogenetic stimulation does not necessitate such physical contact. Thus, the indirect methods provide clear advantage of being non-intrusive. In addition, cellular specificity can be achieved while using optogenetic stimulation. Optogenetic method has been employed for vision restoration in blind mice model either by non-specific stimulation of retina (21) or in a promoter-specific manner including Thyl for RGCs (22-26), mGluR6 targeting ON bipolar cells (27, 28).
The earlier approaches for restoration of vision by optogenetic stimulation of retinal cells use opsins such as ChR2 (21) and others, which requires light intensities order of magnitude higher than ambient lighting conditions. Therefore, clinical success of such opsin molecules in ambient environment for vision restoration is not yet achieved. Further, use of external light source or device (e.g. LED array (29)) to activate such opsins can substantially damage the retinal cells in long-term usage. Therefore, effective optogenetic vision restoration at ambient light level has not been shown yet. By photosensitizing higher order retinal neurons (e.g. bipolar cells) with ambient light-sensitive ion-channel proteins (MCO-010), delivered via safe viral vectors, we aim to restore light sensitivity of retina and thus vision lost due to degenerative diseases.
Methods :
The study was conducted in accordance with ICH and GCP guidelines and approval from institutional ethical committee was obtained before commencement of the study. Informed consent was taken from all study subjects in accordance with guidelines of Declaration of Helsinki.
The study included all the cases presenting to a tertiary care eye hospital in Eastern India with advanced RP after preliminary screening to fit into to the inclusion and exclusion criteria as noted below
Inclusion Criteria
- Age > 18 years
- Ability to comply with testing and all protocol tests.
- Diagnosis of advanced RP based on
- Clinical diagnosis and fundus photography
- Prior documented (if any) retinal electrophysiological evidence of rod-cone photoreceptor degeneration
- Snellen’s visual acuity equivalent LP/NLP in worse (study) eye
- Visual acuity in the non-study eye of no-better-than finger counting
- Presence of retinal bipolar cells and retinal nerve fiber layer on OCT testing
- Women of childbearing potential must have a negative pregnancy test at the screening
- Males must use effective forms of contraception during the study period
Exclusion Criteria
- Participation in a clinical study (ocular or non-ocular) with an investigational drug, agent or therapy in the past six months.
- Concurrent participation in another interventional clinical ocular study.
- Prior participation in any gene or stem cell therapy (ocular or non-ocular).
- Pre-existing eye conditions that would preclude the planned treatment (i.e. injection) or interfere with the interpretation of study endpoints or surgical complications (example would include, but not limited to, glaucoma, diseases affecting the optic nerve causing significant visual field loss, active uveitis, corneal or lenticular opacities).
- Complicating systemic diseases or clinically significant abnormal baseline values.
- Subjects with any immunological response dysfunction, for example, immuno-compromising diseases or use of immunosuppressive medications, among others. Subjects who are positive for hepatitis B, C, and HIV will be excluded.
- Cataract surgery, intraocular and/or peri-ocular injection in the study eye within the prior three months.
- Opacity of lens >3+ due to cataract or significant media opacities hindering visualization of fundus or performance of OCT in the study eye.
- Known sensitivity to any component of the study agent or medications planned for use in the peri-operative period.
- Current pregnancy or breastfeeding.
- Subjects will be excluded if immunological studies show presence of neutralizing antibodies to AAV2 above 1:1000.
- Any other condition that would not allow the potential subject to complete follow-up examinations during the course of the study and, in the opinion of the investigator, makes the potential subject unsuitable for the study.
- Presence of narrow iridocorneal angles contraindicating pupillary dilation.
- Presence of any macular pathology causing decrease in vision or retinal detachment involving macula.
- Active ocular inflammation or recurrent history of idiopathic or autoimmune associated uveitis.
The investigational product is AAV2 carrying Multi Characteristic Opsin ( vMCO1) which have obtained orfan drug status by USFDA. The drug needs to be kept between minus 70 to minus 80 degree centigrade throughout the transportation & storage.
This phase I/IIa clinical study is an open label- dose exploration and expansion in which 3 subjects received low dose (2.5 E11 vg/eye) uniocular intravitreal injection of vMCO-I (in the worst eye). Upon confirming the safety of the low dose, 3 more subjects received the high dose (5.0 E11 vg/eye). Once it was confirmed that the high dose subjects showed safety and tolerability, 5 more subjects received the high dose of 5E11 vg/eye through intravitreal administration. The subjects were regularly monitored and followed up according to the schedule mentioned on day 1, 2nd, 4th, 8th ,12th, 16th & 52nd week. (Fig. – 1)
The objectives of this Phase I/IIa clinical study is to:
- Evaluate the safety and tolerability of the intravitreal administration of adeno-associated virus, serotype 2 (AAV2) carrying ambient light activatable Multi Characteristic Opsin (vMCO-I) in patients with advanced RP.
- To define the safety (Phase I) and confirm highest tolerated dose (Phase IIa) and recommend Phase IIb dose
Data was collected on a standardised form, which included age, sex, address, presenting vision, ophthalmic examination, investigations, number of follow ups, sequelae, complications and final visual outcome. A detailed history was taken. All subjects underwent comprehensive ophthalmologic examination. Complete external examination was done by slit lamp biomicroscopy, intraocular pressure (IOP) was measured by using rebound tonometer (I-care). Fundus was evaluated using 90 D Volk lens and by indirect ophthalmoscopy with 20D lens. Screening for pregency, HIV, HBSAg, fasting blood sugar (FBS), postprandial blood sugar (PPBS), complete blood count (CBC), lipid profile and renal profile were done for every patient.
Under aseptic conditions, patients underwent intravitreal injection of vMCO1 in the operating room under topical anaesthesia using 30G needle in a micro titrated syringe. Subjects were on an oral regimen of systemic corticosteroids beginning three days before the administration of vMCO-I (Day -3). The initial dose was 1 mg/kg/day prednisone for seven days, with a maximum prescribed dose of 40 mg/day, regardless of the weight of the subject; this was followed by 0.5 mg/kg/day prednisone for an additional five days, with a maximum prescribed dose of 20 mg/day, regardless of the weight of the subject. Subjects were on systemic corticosteroids for a minimum of 12 days up to a maximum of 30 days, depending on the post-injection inflammation.
Subjects with mild inflammation received topical steroids or periocular/sub-Tenon/intravitreal triamcinolone acetonide at the discretion of the investigator. Some subjects were treated for increase in intraocular pressure with IOP lowering medicines.
Subjects underwent recording of Adverse Events as reported by participant or observed by investigator Recorded concurrent medications, Recorded vital signs, weight, Physical examination, Functional assessment of vision by Electro Retinogram (ERG), Visual performance status by Low Vision Multi parametric test (LVMPT), Full-field light sensitivity threshold, Fundus photography, Visually-guided behavioral assays, Performed OCT imaging, record results, Visual function questionnaire, Best-corrected visual acuity (Freiburg ACT), Humphrey Visual Field in each visit. Blood drawn (for Neutralizing Antibody assay and Complete Blood Count on 4th week and for genotyping on 8th week.
All data were analysed using SPSS software version 24.42 (IBM SPSS Statistics 24.42, www.spss.co.in, India) for windows. Statistical analysis was done and significance of correlation for each parameter was calculated using Chi- square test and descriptive statistics. Percentages and frequencies were calculated for demographic variables as well as clinical parameters. Tables and graph was used to present the results.
Results:
The totality of data from clinical development program demonstrates that vMCO-I has positive safety profile at the dose of 5.0E11vg/eye. The study also demonstrated the evidence of efficacy at the end of 16 weeks after intravitreal injection. Improvements in both visual function and functional vision was reported as early as 4 weeks which further improved through 16 weeks. There was no major AEs reported upon administration of vMCO-I. An interesting observation that was visible during this study was the presence of MCO-010 reporter (mCherry) in the contralateral eyes of some patients which led to improvement in the contralateral eye too. The logMAR acuity value of injected eyes in the high-dose group showed improvement of 0.68 as compared to 0.1 logMAR in low-dose group 16 weeks after injection. The mean improvement in the logMAR acuity in high-dose contralateral eyes was found to be 0.34 logMAR at 16 weeks as compared to improvement of 0.68 logMAR acuity in the injected eyes. Dose-dependent improvement in FST-value was observed in both injected and contralateral eyes. An increased visual field index in 302 Humphrey visual assessment was observed in both injected and contralateral eyes for few patients.
Longitudinal measurement using LVMPT showed that the intensity threshold to detect different static shapes correctly reduced after vMCO-010 injection. Further, at the threshold intensity level (22 Lux), the shape (circle, triangle and square) determination accuracy increased from ~50% at baseline to 100% at >8 weeks for both injected and contralateral eyes. Similar test for determining the Size threshold for detecting different shapes reduced to half after vMCO-010 injection implying halving of visual angle. In addition, longitudinal measurement using LVMPT showed that the accuracy (%) in detection of direction of Optical Flow increased from ~40% at baseline to ~100% at >8 weeks for both vMCO-010 injected and contralateral eyes. Furthermore, the upper speed threshold (to accurately detect direction of optical flow) increased significantly (mean value more than doubled) 8 weeks after vMCO-010 injection.
For both Y- and A-Mobility tests, the mean latency and standard error around mean to find lighted panel is decreased significantly after vMCO-10 injection as compared to the baseline. Light-intensity dependent improvement was observed in A-Mobility assay with the mean latency and standard error around mean for 1 lux assay was found to be lower than that at 0.5 lux at > 4 weeks after vMCO-010 injection. The Mobility scores in both Y- and A-Mobility tests improved after vMCO-010 injection and this improvement was observed bilaterally. Overall, all the subjects in all trials made less errors and performed the tasks faster after vMCO-010 injection irrespective of presence (A-maze) or absence (Y-maze) of obstacles, position of the lighted LED panel (left or right), starting positions (middle or off-centered), or intensity levels (0.5, 1 lux) of the lighted LED panel.
The overall composite score and the three subscales (near activities, distance activities, and vision-specific dependency) showed significant improvement at 4, 8 and 16 weeks after injection. The increasing trend of the scores was associated with the increase in the measured visual acuity and other visual functions, namely light stimulation threshold, visual field, shape and direction recognition ability, visually guided mobility etc. Vision-specific dependency subscale score increased the most among the other subscale scores. This increased subscale score correlated with anecdotes from the subjects and their family members.
A comprehensive ophthalmic assessment with dilated fundus examination was performed to grade the magnitude of the inflammation by compartment and severity, according to the SUN criteria for anterior chamber (AC) cell and flare (30), and the NEI grading scheme for vitreous haze (30, 31).
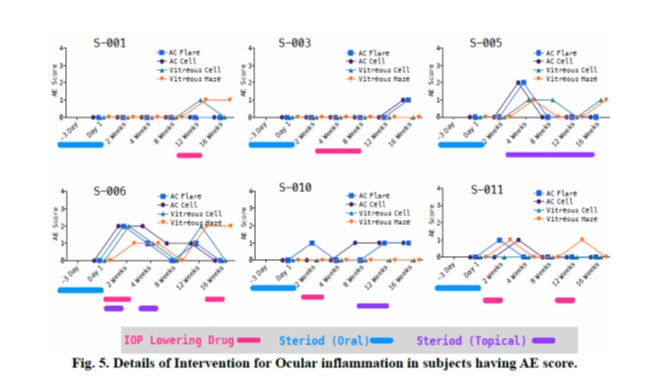
Mild to moderate ocular inflammation and increase in IOP in some subjects were observed and were treated with topical steroid and/or IOP-lowering drops (Fig. 2). S-003 (low dose) and S-006 (high dose) had moderate increase in IOP level after injection. Few other subjects had mild increase in IOP, which was controlled via application of topical steroid/IOP-lowering drug without requiring surgery. At 16 weeks, the average IOP level maintained similar to normal values. To evaluate if elevated IOP was related to vMCO-010 injection related intraocular inflammation, ocular adverse events were evaluated by slit lamp and indirect ophthalmoscopy.
No subject had an ocular inflammation score greater than 2 at any timepoint during the study. At 2 weeks after injection, 1 subject (S-006) had AC/vitreous AE grade 2 and 2 subjects (S-010, 011) had AC/vitreous AE Grade of 1. At 4 weeks, 2 subjects (S-005, 006) had AC/vitreous AE grade 2/1 and 1 subject (S-011) had AC/vitreous AE Grade of 1. At 8 weeks of injection, only 1 subject (S-005) had AE Grade of 1 in AC/Vitreous. 5 subjects (S-001, 002, 009, 010, 011) had AE score Grade of 1 in AC/Vitreous at 12 weeks after injection and 1 subject (S-006) has AC/Vitreous AE grade 1/2. At 16 weeks, 1 subject (S-006) had vitreous Haze AE grade 2; 5 subjects (S-001, 003, 004, 005, 008) had AC/ vitreous AE Grade of 1. An ocular AE (Keratic precipitate) in 1 subject (S-005) leading to corneal endothelial deposit could not be confirmed to be related to intravitreal injection. Fig. 3 shows the longitudinal assessment of Anterior Chamber Cell and Anterior Chamber Flare in the injected and contralateral eyes. In Fig. 4, we show longitudinal assessment of Vitreous Cell and Vitreous haze in injected and contralateral eye and Fig. 5 shows the details of the intervention for ocular inflammation in subjects having AE score > 0.
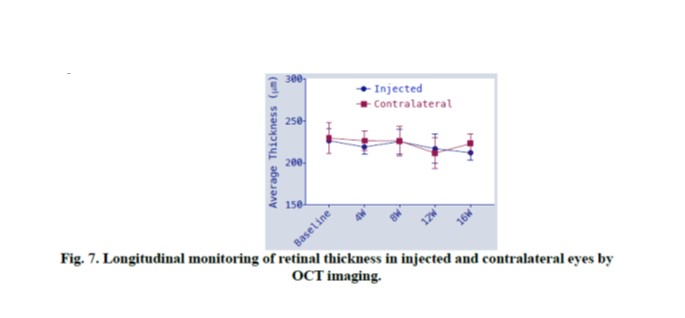
Retinal thinning is known to occur in RP due to progressive nature of the degeneration. To monitor the rate of retinal thinning and to evaluate retinal thinning (if any) due to vMCO-I injection, OCT imaging of retina was carried out in a longitudinal manner. In Fig. 6, we have shown OCT images of injected eye at Baseline and 16 weeks after injection in 2 low dose and 2 high dose subjects having ocular inflammation. Longitudinal assessments of retinal thickness in these subjects (Fig. 6) show no dependence on administered dose or change in IOP.
Longitudinal monitoring of retinal thickness in injected and contralateral eyes was carried out by OCT imaging and quantified (Fig. 7). Measured average retinal thickness did not change after vMCO-010 injection. No significant change in retina thickness was observed at 16 weeks with respect to baseline.
The vMCO-010 safety profile remains satisfactory and MCO-010 was well-tolerated 16 weeks after intravitreal injection. No serious adverse events observed in vMCO-010 injected eyes or contralateral eyes and all dosed subjects continue active participation. Mild to moderate ocular inflammation and increase in IOP in some subjects was treated with topical steroid and/or IOP-lowering drops. There was no itching, pain or redness of eye reported by the subjects. Furthermore, no significant change in retina thickness was observed at 16 weeks with respect to baseline as measured by OCT. The analysis of AAV2 neutralizing antibody 4 weeks after injection shows no detectable increase suggesting no systemic immune response.
Discussion
MCO-010 is an ambient light-activatable ion channel protein, which opens up when exposed to light allowing flow of cations into the cell. Our in-vivo animal results show significant photo-induced inward current upon activation of MCO-010 expressing cells by low intensity of light. vMCO-I is MCO-010 carried by adeno-associated virus serotype 2 (AAV2), which is naturally replication deficient, requiring co-infection with helper viruses to replicate. The wild-type virus consists of a single-stranded DNA genome encapsulated in a protein coat. The genome consists of three elements: the rep gene, the cap gene, and the inverted terminal repeats (ITRs). The rep gene codes for proteins involved in DNA replication, and the cap gene, which, through a differential splicing mechanism, encodes three amino-terminal variant virus proteins, VP1, VP2 and VP3, that make up the coat of the virus.
In order to achieve optogenetic stimulation of retinal neurons, the retinal cells especially the ON bipolar cells are generally transfected by a vMCO-I (administered intravitreally) to express multi-characteristic opsin (light-sensitive molecular ion-channel), which gets activated, thus depolarizing the opsin-expressing retinal bipolar cells when illuminated by ambient light in broad visible spectrum (characteristics of the multi-characteristic opsin). The photosensitized bipolar cells have shown to drive retinal circuitry functions, activate cortical circuits, and mediate visually guided behaviors.
The doses and injection volume have been calculated by allometric scaling from pre-clinical animal studies. Vitreous humor volume was the scaling factor across species. In general, scaling of the vitreous volume from mice to humans is 1: 1000. In our preclinical studies, we have conducted biodistribution analysis in non-targeted tissues (using quantitative PCR) and toxicology studies (i.e. pro-inflammatory cytokine quantification using ELISA, apoptotic biomarker expression in retina) in rd10 mice after vMCO-I injection. We have used intravitreal dose up to 1.0 E10 vg/eye in mouse and found it to be safe as evaluated by OCT for ocular structures, ELISA for inflammatory cytokines, QPCR for biodistribution and immunostaining for determining immune cell response. The GLP toxicity studies (Ophthalmic examinations, ERG, IOP, Body weight, Tear test, ELISA, Clinical Pathology, Histopathology, Biodistribution and immunohistochemistry) on wild type dogs conducted by CRO (Contract Research Organization) did not show limiting toxicity for dose up to 0.64 E12vg/eye. Therefore, the expected safe dose limit for vMCO-I in human is estimated (based on ratio of dog to human eye vitreous volume being ~1:3) to be 1.92 E12 vg/eye of vMCO-I.
The proposed route of administration of vMCO-I is via single uniocular intravitreal injection. This is based on following: (i) intravitreal injection is a well-established minimally-destructive method for drug delivery to retina; (ii) anatomically isolated intravitreal delivery minimizes circulation of vMCO-I in other organs (as evidenced by the biodistribution study); (iii) Single uniocular injection further minimizes risks (if any) of inflammation in non-injected eye.
The vMCO-010 safety profile remains satisfactory and MCO-010 was well-tolerated 16 weeks after intravitreal injection. No serious adverse events observed in MCO-010 injected eyes or contralateral eyes and all dosed subjects continue active participation. Mild to moderate ocular inflammation and moderate increase in IOP in some subjects was treated with topical steroid and/or IOP-lowering drops. There was no itching, pain or redness of eye reported by the subjects. Furthermore, no significant change in retina thickness was observed at 16 weeks with respect to baseline as measured by OCT. The analysis of AAV2 neutralizing antibody 4 weeks after injection shows no detectable increase suggesting no systemic immune response.
Dose-response evidence of efficacy of vMCO-I through 16 weeks was observed including: (i) Improvement of outdoor light sensitivity and daily activities (ii) Improvements in visual acuity (iii) Decrease in light stimulation threshold (FST) (iv) increased visual field index in 30-2 Humphrey visual assessment (iv) decreased latency and improved score in Y and A-Mobility test (v) lowering of intensity/size threshold as well as increase in accuracy of shape/direction detection and upper speed threshold for optical flow in Low Vision Multi-Parameter Test (LVMPT) and (vi)improvement in NEI-VFQ overall composite score as well as the three subscales scores on near activities, distance activities, and vision-specific dependency.
Conclusions:
The overall preliminary safety and efficacy results from the phase I/IIa clinical study demonstrated that the benefit risk balance is strongly in favor vMCO-I for the treatment of patients with vision loss due to RP. Intravitreal Optogenetic Gene Therapy in patients of Retinitics Pigmentosa with vMCO-l is well tolerated with no serious adverse events. Higher dose vMCO-l appears to improve visual acuity and visual function at 16 weeks compared to baseline measurements and demonstrates tremendous promise in restoring vision for retinitis pigmentosa patients.
- Hartong DT, Berson EL, Dryja TP. Retinitis pigmentosa. Lancet. 2006;368(9549):1795-809.
- Sugawara T, Hagiwara A, Hiramatsu A, Ogata K, Mitamura Y, Yamamoto S. Relationship between peripheral visual field loss and vision-related quality of life in patients with retinitis pigmentosa. Eye (Lond). 2010;24(4):535-9.
- Daiger SP, Bowne SJ, Sullivan LS. Perspective on genes and mutations causing retinitis Arch Ophthalmol. 2007;125(2):151-8.
- Mezer E, Babul-Hirji R, Wise R, Chipman M, DaSilva L, Rowell M, et al. Attitudes Regarding Predictive Testing for Retinitis Pigmentosa. Ophthalmic Genetics. 2007;28(1):9-15.
- Flannery JG, Farber DB, Bird AC, Bok D. Degenerative changes in a retina affected with autosomal dominant retinitis pigmentosa. Invest Ophthalmol Vis Sci. 1989;30(2): 191-211.
- Hartong DT, Berson EL, Dryja TP. Retinitis pigmentosa. Lancet. 2006;368(9549):1795-809.
- Li ZY, Jacobson SG, Milam AH. Autosomal dominant retinitis pigmentosa caused by the threonine-17-methionine rhodopsin mutation: retinal histopathology and immunocytochemistry. Exp Eye Res. 1994;58(4):397-408.
- Grover S, Fishman GA, Anderson RJ, Alexander KR, Derlacki DJ. Rate of visual field loss in retinitis pigmentosa. Ophthalmology. 1997;104(3):460-5.
- Baumgartner WA. Etiology, pathogenesis, and experimental treatment of retinitis pigmentosa. Medical Hypotheses. 2000;54(5):814-24.
- Sahaboglu A, Paquet-Durand O, Dietter J, Dengler K, Bernhard-Kurz S, Ekstrom PA, et al. Retinitis pigmentosa: rapid neurodegeneration is governed by slow cell death mechanisms. Cell Death Dis. 2013;4:e488.
- Hamel C. Retinitis pigmentosa. Orphanet J Rare Dis. 2006; 1:40.
- Xia Y, Peng X, Ren Q. Retinitis pigmentosa patients’ attitudes toward participation in retinal prosthesis trials. Contemp Clin Trials. 2012;33(4):628-32.
- Yanai D, Weiland JD, Mahadevappa M, Greenberg RJ, Fine I, Humayun MS. Visual performance using a retinal prosthesis in three subjects with retinitis pigmentosa. Am J Ophthalmol. 2007;143(5):820-7.
- Kusnyerik A, Greppmaier U, Wilke R, Gekeler F, Wilhelm B, Sachs HG, et al. Positioning of electronic subretinal implants in blind retinitis pigmentosa patients through multimodal assessment of retinal structures. Invest Ophthalmol Vis Sci. 2012;53(7):3748-55.
- Horsager A, Greenwald SH, Weiland JD, Humayun MS, Greenberg RJ, McMahon MJ, et al. Predicting visual sensitivity in retinal prosthesis patients. Invest Ophthalmol Vis Sci. 2009;50(4):1483-91.
- de Balthasar C, Patel S, Roy A, Freda R, Greenwald S, Horsager A, et al. Factors affecting perceptual thresholds in epiretinal prostheses. Invest Ophthalmol Vis Sci. 2008;49(6):2303-14.
- Zrenner E, Bartz-Schmidt KU, Benav H, Besch D, Bruckmann A, Gabel VP, et al. Subretinal electronic chips allow blind patients to read letters and combine them to words. Proc Biol Sci. 2011;278(1711): 1489-97.
- Chow AY, Pardue MT, Perlman JI, Ball SL, Chow VY, Hetling JR, et al. Subretinal implantation of semiconductor-based photodiodes: durability of novel implant designs. J Rehabilit Res Develop. 2002;39(3):313-21.
- Zrenner E. Will Retinal Implants Restore Vision? Science. 2002;295(5557):1022-5.
- Eckmiller R. Learning Retina Implants with Epiretinal Contacts. Ophthal Res. 1997;29(5):281-9.
- Bi AD, Cui JJ, Ma YP, Olshevskaya E, Pu ML, Dizhoor AM, et al. Ectopic expression of a microbial-type rhodopsin restores visual responses in mice with photoreceptor degeneration. Neuron. 2006;50(1):23-33.
- Thyagarajan S, van Wyk M, Lehmann K, Lowel S, Feng G, Wassle H. Visual Function in Mice with Photoreceptor Degeneration and Transgenic Expression of Channelrhodopsin 2 in Ganglion Cells. J Neurosci. 2010;30(26):8745-58.
- Bi A, Cui J, Ma YP, Olshevskaya E, Pu M, Dizhoor AM, et al. Ectopic expression of a microbial-type rhodopsin restores visual responses in mice with photoreceptor degeneration. Neuron. 2006;50(1):23-33.
- Zhang Y, Ivanova E, Bi A, Pan Z-H. Ectopic Expression of Multiple Microbial Rhodopsins Restores ON and OFF Light Responses in Retinas with Photoreceptor Degeneration. J Neurosci.2009;29(29):9186-96.
- Tomita H, Sugano E, Isago H, Hiroi T, Wang Z, Ohta E, et al. Channelrhodopsin-2 gene transduced into retinal ganglion cells restores functional vision in genetically blind rats. Experimental Eye Research. 2010;90(3):429-36.
- Tomita H, Sugano E, Fukazawa Y, Isago H, Sugiyama Y, Hiroi T, et al. Visual Properties of Transgenic Rats Harboring the Channelrhodopsin-2 Gene Regulated by the Thy-1.2 Promoter. PLoS One. 2009;4(11).
- Lagali PS, Balya D, Awatramani GB, Munch TA, Kim DS, Busskamp V, et al. Light-activated channels targeted to ON bipolar cells restore visual function in retinal degeneration. Nat Neurosci. 2008;11(6):667-75.
- Doroudchi MM, Greenberg KP, Liu J, Silka KA, Boyden ES, Lockridge JA, et al. Virally delivered Channelrhodopsin-2 Safely and Effectively Restores Visual Function in Multiple Mouse Models of Blindness. Mol Ther. 2011;19(7):1220-9.
- Degenaar P, Grossman N, Memon MA, Burrone J, Dawson M, Drakakis E, et al. Optobionic vision-a new genetically enhanced light on retinal prosthesis. Journal of neural engineering. 2009;6(3).
- Group SoUNW. Standardization of uveitis nomenclature for reporting clinical data. Results of the First International Workshop. American journal of ophthalmology. 2005;140(3):509-16.
- Nussenblatt RB, Palestine AG, Chan C-C, Roberge F. Standardizatlon of Vitreal inflammatory Activity in Intermediate and Posterior Uveitis. Ophthalmology. 1985;92(4):467-71.








 FP0972 : Phase 1/2 study of intravitreal gene therapy for vision restoration in advanced retinitis pigmentosa
FP0972 : Phase 1/2 study of intravitreal gene therapy for vision restoration in advanced retinitis pigmentosa
Leave a Comment